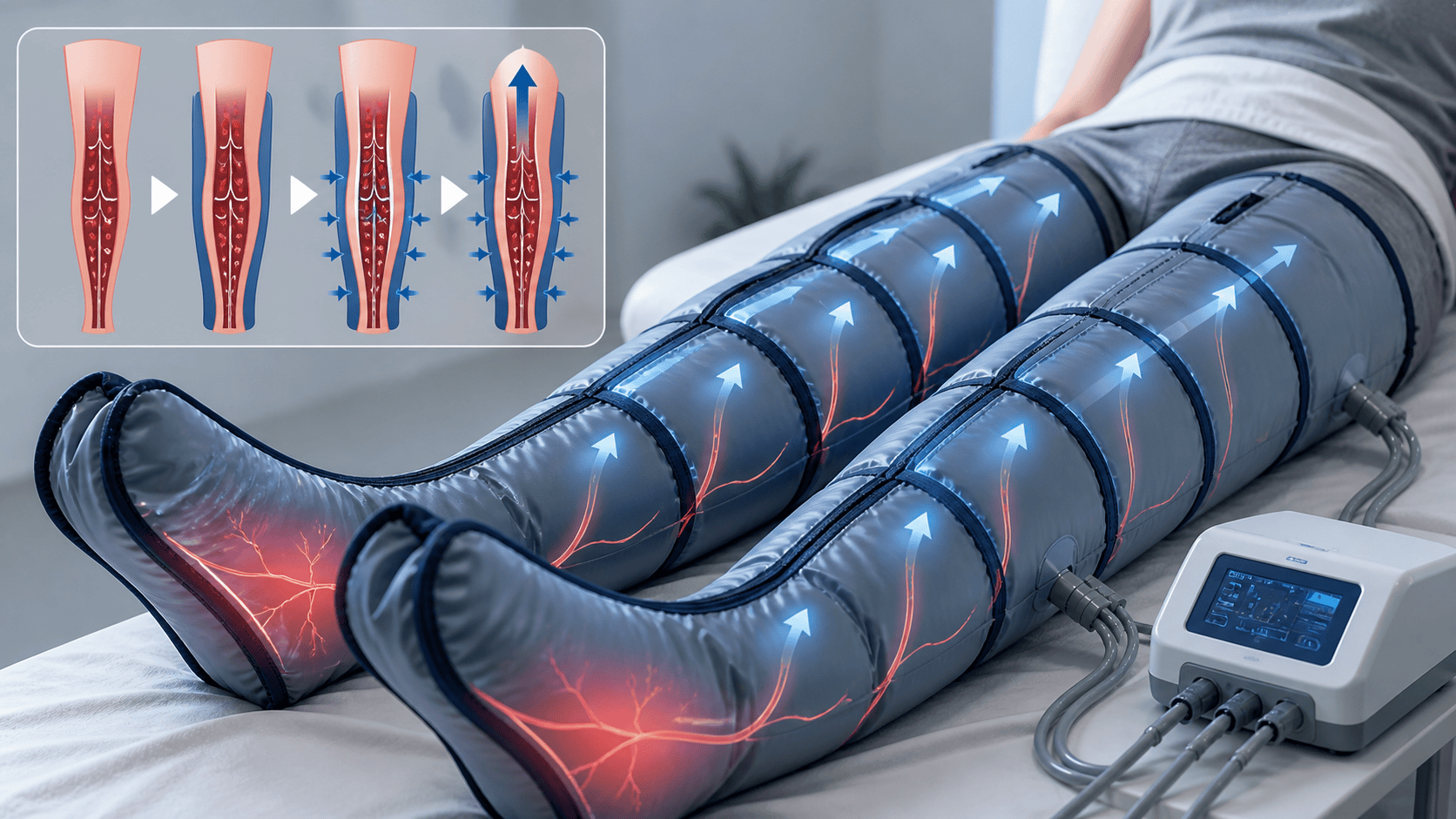
Deep vein thrombosis prevention is where compression boots have their longest and strongest evidence base. Long before these devices appeared in gyms and recovery studios, intermittent pneumatic compression was standard hospital equipment, keeping blood moving in immobilized patients who could not walk. The DVT prevention application is not a secondary benefit or a marketing claim. It is the original reason this technology exists.
That said, there is a critical distinction between clinical-grade pneumatic compression DVT devices used in hospitals and the consumer recovery boots sold for athletic use. They share a mechanism but differ in clearance, design, and intended purpose. Here is what the clinical research actually supports, where consumer boots fit into the picture, and what you need to know if DVT risk is your primary concern.
What DVT Is and Why It Matters
Deep vein thrombosis occurs when a blood clot forms in one of the deep veins, most commonly in the lower legs or thighs. The clot partially or fully blocks blood flow through the vein, causing pain, swelling, warmth, and redness in the affected leg. Some DVTs are silent, producing no symptoms at all.
The serious danger is not the clot itself. The serious danger is pulmonary embolism (PE), which occurs when part of the clot breaks free, travels through the bloodstream, and lodges in the lungs. PE can be fatal. In hospitalized surgical patients, venous thromboembolism (the umbrella term covering both DVT and PE) is one of the most common preventable causes of death.
Three factors drive DVT formation, known collectively as Virchow’s triad: blood flow stasis (blood moving too slowly or pooling), vascular damage (injury to the vein wall), and hypercoagulability (blood that clots more easily than normal). Surgery, immobilization, and certain medical conditions trigger one or more of these factors.
Compression boots address the first factor directly. By mechanically pushing venous blood upward from the legs, they prevent the stasis that allows clots to form in the first place.
How Intermittent Pneumatic Compression Prevents Clots
The mechanism is straightforward and well understood. IPC devices inflate air chambers sequentially from foot to thigh, compressing the deep veins and forcing blood upward through one-way venous valves toward the heart. When the chambers deflate, the veins refill with fresh blood. The cycle repeats, maintaining continuous blood flow movement even in a patient who is completely immobile.
This replicates the calf muscle pump, the body’s primary mechanism for moving venous blood against gravity. When you walk, calf muscle contractions compress the deep veins and push blood upward. When you lie in a hospital bed after surgery, sit for 12 hours on a long-haul flight, or are immobilized by a stroke, that pump stops working. The boots substitute mechanical compression for the missing muscle contractions.
Beyond simply moving blood, IPC also appears to have a secondary effect on the coagulation system itself. Some research suggests that the pulsatile compression increases release of tissue plasminogen activator (tPA), a natural clot-dissolving enzyme, from the vessel walls. This anticoagulant effect supplements the mechanical flow enhancement, though the clinical significance of this secondary pathway is less firmly established than the primary flow effect.
The Clinical Evidence for DVT Prevention
The evidence base for deep vein thrombosis boots in hospital settings is extensive, spanning decades of randomized trials across multiple patient populations.
Surgical patients: a 2024 systematic review and meta-analysis in PLOS ONE analyzed 16 randomized controlled trials and found that IPC significantly reduced DVT incidence compared to no prophylaxis, with an odds ratio of 0.41 (meaning IPC cut DVT risk by roughly 59% compared to patients receiving no prevention). The same analysis found that IPC also significantly reduced bleeding risk compared to pharmacological prophylaxis combined with IPC, making it particularly valuable for patients where blood thinners carry excessive bleeding risk.
Stroke patients: the landmark CLOTS 3 trial, published in The Lancet in 2013, randomized 2,876 immobile stroke patients across 105 UK centers. The trial found that IPC significantly reduced the risk of proximal DVT, symptomatic DVTs, and all DVTs compared to standard care alone. The CLOTS 3 collaboration also reported that IPC was associated with improved survival. This was the largest trial of IPC in non-surgical patients and changed clinical practice in many stroke units.
Gynecologic surgery patients: a meta-analysis of 7 randomized controlled trials covering 1,001 patients found that IPC significantly lowered DVT risk compared to control, with a risk ratio of 0.33 (a 67% relative risk reduction). Notably, the DVT incidence with IPC was comparable to pharmacological prophylaxis, but IPC had a lower postoperative transfusion rate, meaning fewer bleeding complications.
Critically ill patients: a meta-analysis of IPC in ICU populations found that it could reduce VTE risk without increasing bleeding events or all-cause mortality, an important finding because critically ill patients are often at high risk for both clots and bleeding simultaneously.
Trauma patients: a 2023 evidence-based review summarizing 19 studies (including 12 guidelines, 4 systematic reviews, and expert consensus documents) confirmed IPC as a recommended prophylactic intervention for trauma patients, with 27 evidence-based recommendations covering evaluation, contraindications, treatment strategies, and patient education.
Clinical IPC Devices vs Consumer Compression Boots
This distinction is the most important one in this article, and it is the one most often blurred by marketing.
Clinical-grade IPC devices used in hospitals (Kendall SCD, Covidien, Cardinal Health systems) are FDA-cleared Class II medical devices specifically designed and validated for DVT prophylaxis. They typically feature rapid-inflation sequential compression, venous refill time sensors that adjust cycle frequency to the individual patient, calf-length or thigh-length configurations validated in clinical trials, and compliance monitoring that alerts nursing staff when the device is removed.
Consumer recovery boots (Normatec, Therabody, Fit King, Rapid Reboot) are designed for athletic recovery and wellness. They use the same core mechanism of sequential pneumatic compression, but they are not FDA-cleared for DVT prevention, have not been validated in DVT prevention trials, and lack the rapid-inflation timing and compliance features of clinical devices.
The core mechanism is identical: sequential compression that pushes venous blood upward. But the clinical validation is not. No consumer recovery boot has been tested in a randomized trial for DVT prevention. The evidence supporting IPC for DVT prevention comes from clinical-grade devices used under medical supervision in hospital settings.
This does not mean consumer boots have zero DVT-relevant benefit. The venous return effect is real and measurable regardless of the brand name on the device. But no one should rely on a consumer boot as their primary DVT prophylaxis in a clinical situation where a validated medical device and/or pharmacological prophylaxis is indicated.
Where Consumer Boots May Help With DVT-Related Risk
While consumer boots are not validated for DVT prevention in clinical contexts, there are everyday situations where their venous return benefit is relevant to DVT risk factors.
Long-haul travel. Extended immobility during flights, trains, and car trips is a recognized DVT risk factor. Consumer boots used before departure or after arrival can help restore venous flow after hours of sitting. They do not replace compression socks worn during the flight or the practice of walking the aisle every hour, but they supplement those measures.
Sedentary work patterns. Desk workers and drivers who sit for 8+ hours a day face chronic venous stasis that accumulates over years. Regular use of consumer boots in the evening addresses the daily fluid pooling that prolonged sitting creates. This is a wellness and comfort benefit, not a clinical DVT prevention protocol, but it addresses the same underlying stasis problem.
Post-travel recovery. Using consumer boots after arriving at your destination helps clear the venous pooling that built up during the journey. This is particularly useful after international flights of 8+ hours, where the combination of cabin pressure, dehydration, and immobility creates significant lower-leg swelling.
In all of these situations, the boots are a comfort and wellness tool that supports venous return, not a medical device preventing a diagnosed clinical risk. If your physician has told you that you are at elevated DVT risk due to a specific medical condition, surgical history, or coagulation disorder, follow their prescribed prophylaxis protocol, which may include clinical-grade IPC, anticoagulation, and/or medical-grade compression stockings.
Who Is at Elevated DVT Risk
Understanding the risk factors helps contextualize when the clinical evidence applies and when consumer-level compression is sufficient.
High-risk situations that typically warrant clinical DVT prophylaxis: major surgery (especially hip, knee, or abdominal), hospitalization with immobility, acute stroke with immobility, active cancer, and known clotting disorders (Factor V Leiden, protein C or S deficiency, antiphospholipid syndrome). In these situations, clinical-grade IPC, pharmacological prophylaxis, or both are the standard of care.
Moderate-risk situations where general venous support is reasonable: long-haul flights (4+ hours), prolonged bed rest at home, sedentary work with minimal movement, oral contraceptive use (which increases clotting risk), pregnancy (especially third trimester with limited mobility), and family history of DVT. In these situations, compression socks, hydration, regular movement, and consumer compression boots are all reasonable general measures.
Lower-risk situations where standard health habits are sufficient: generally healthy, active adults who move regularly throughout the day. For this group, DVT risk is low and no specific prophylaxis is needed beyond staying active, hydrated, and avoiding prolonged immobility.
The Contraindication That Matters Most
This is the single most important safety point in this article. If you already have an active DVT or an unstable blood clot, compression boots of any kind, clinical or consumer, are absolutely contraindicated.
The sequential compression that prevents clots from forming can also dislodge a clot that has already formed. If a piece of the clot breaks free and travels to the lungs, the result is a pulmonary embolism, which can be life-threatening.
Johns Hopkins Medicine, the American College of Chest Physicians, and every major vascular guideline list active DVT as a contraindication for IPC. Prevention and treatment are opposite clinical situations. The boots prevent clots by keeping blood moving. They cannot treat existing clots and can make them dangerous.
If you develop sudden leg swelling, pain, warmth, or redness, especially in one leg, do not use compression boots. Seek medical evaluation immediately.
Compression Boots for DVT Prevention, Frequently Asked Questions
Do hospital compression boots actually prevent blood clots?
Yes. The evidence is extensive and strong. A systematic review of 22 randomized trials found a 64% reduction in DVT odds with IPC, and the CLOTS 3 trial of 2,876 stroke patients demonstrated significant DVT reduction and improved survival. IPC is standard care in most hospitals for immobilized patients precisely because the evidence supporting it is robust.
Can I use Normatec boots to prevent DVT during travel?
Consumer boots like the Normatec produce measurable venous return improvements, which addresses the stasis component of DVT risk during travel. However, they are not FDA-cleared for DVT prevention and have not been tested in DVT prevention trials. For travel DVT risk, they are a reasonable comfort supplement alongside compression socks, hydration, and regular movement, but they are not a validated prophylactic device.
How long do hospital patients wear compression boots?
Clinical protocols vary, but most hospitals apply IPC devices continuously (day and night) from admission through mobilization. In the CLOTS 3 trial, thigh-length IPC sleeves were applied from randomization through Day 30 while patients were sitting or lying down. The devices are typically removed only for bathing, skin inspection, and once the patient is walking independently.
Are clinical compression boots the same as recovery boots?
The core mechanism is identical: sequential pneumatic compression that pushes blood upward. The clinical validation is different. Hospital devices are FDA-cleared for DVT prevention, feature rapid-inflation timing and compliance monitoring, and have been tested in randomized trials with thousands of patients. Consumer boots are designed for athletic recovery and are not validated for DVT prevention.
Should I use compression boots if I have a history of blood clots?
Only with your vascular specialist’s clearance. If you have had a DVT in the past but are not currently experiencing active clotting, compression therapy may be safe and even beneficial once anticoagulation is managed. But the decision belongs to your physician, not to a consumer product recommendation. Active DVT remains an absolute contraindication for any compression device.
Do compression socks work as well as boots for DVT prevention?
For different purposes. Graduated compression stockings provide continuous passive support throughout the day and are well-studied for travel DVT risk and chronic venous insufficiency. IPC boots provide active sequential compression during short sessions. In hospital settings, the two are sometimes used together. For everyday DVT risk management, compression socks worn during prolonged sitting are more practical than boots, since you can wear them during a flight rather than after.
Can compression boots cause a pulmonary embolism?
In a person with an existing unstable blood clot, compression could theoretically dislodge the clot and cause a PE. This is why active DVT is an absolute contraindication for compression devices. In people without existing clots, the boots reduce PE risk by preventing clot formation in the first place. The CLOTS 3 trial specifically confirmed this preventive benefit in immobile stroke patients.
The Bottom Line
Intermittent pneumatic compression is one of the most effective non-pharmacological tools for DVT prevention, backed by decades of clinical trials across surgical, stroke, trauma, and critically ill patient populations. The mechanism is well established, the risk reduction is significant, and the technology is standard hospital care worldwide.
Consumer compression boots share the same sequential compression mechanism and produce measurable venous return improvements, but they are not validated for DVT prevention and should not be treated as a medical prophylaxis device. They are a reasonable comfort and wellness supplement for everyday venous stasis from travel and sedentary work, used alongside compression socks, hydration, and regular movement.
If you have a diagnosed DVT risk factor, a history of blood clots, or are facing surgery that increases clot risk, follow your physician’s prophylaxis protocol. That may include clinical-grade IPC, anticoagulation, graduated compression stockings, or a combination. Consumer boots can supplement general vascular health but cannot replace clinical-grade prevention.